All systems move in the direction of increasing entropy.Thus Clausius introduced the concept of entropy in the middle of the 19th century. 1.1 Entropy in Thermodynamics and Statistical Mechanics. Thus, entropy has the units of energy unit per Kelvin, J K -1. Entropy is the amount of energy transferred divided by the temperature at which the process takes place. 
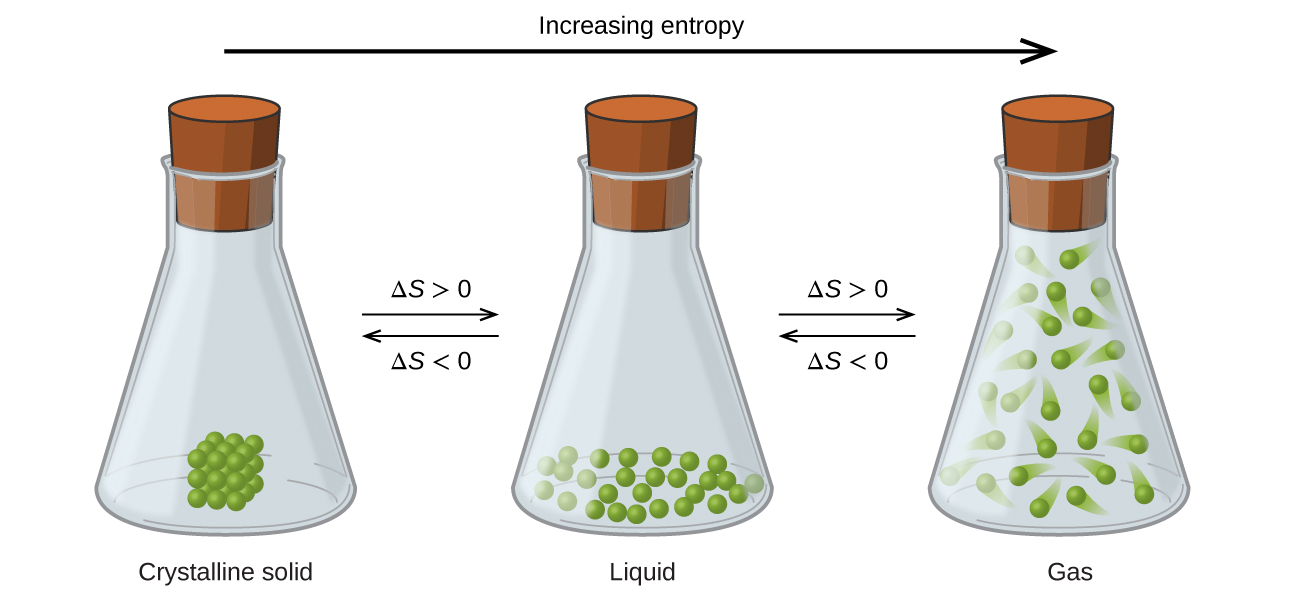
The solid phase has lower entropy, whereas the liquid phase has higher entropy. When a system receives an amount of energy q at a constant temperature, T, the entropy increase D S is defined by the following equation. When in a reaction the number of moles of product formed is greater than the number of moles of reactants, there is an increase in entropy.Īn entropy change will be negative when the opposite changes are seen. Phil Attard, in Thermodynamics and Statistical Mechanics, 2002. There is a change in the entropy when there is a change in the phase transition. For an increased number of particles there is and increased number of ways in which these can be arranged and therefore an increased entropy. 
The arrangement of particles is more random in gases than either in solids or liquids.Ģ. Before carrying out any calculations to find out its exact value, we can infer the direction that this entropy change go in by simply comparing reactants and products.ġ. When energy is transferred from a hotter object to a colder one, some of. In a chemical reaction, an entropy change takes place as the reactants are changed into products. The increase in entropy is related to the concept of heat transfer and energy flow. The more disordered a system is, the higher (the more positive) the value of entropy. Thermodynamic - Entropy Change Predictions (A-Level Chemistry) Entropy Change Predictions Predicting Entropy ChangesĪs we have already explained, entropy can be defined as the randomness or dispersal of energy of a system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
